The Bordetella species, B. pertussis, B. parapertussis, and B. bronchiseptica, the so-called classical bordetellae, cause respiratory infections in diverse mammals, although they differ in their host range and disease manifestations.
B. pertussis is a strictly human-adapted pathogen that lacks an animal reservoir and cannot survive outside the host. It is the causative agent of whooping cough, also known as pertussis, a severe and sometimes fatal respiratory disease, especially in unvaccinated infants. Despite the availability of vaccines, pertussis remains a major public health concern worldwide.
In contrast to B. pertussis, B. bronchiseptica primarily infects animals, such as pigs, dogs and cats, and can persist in environmental reservoirs. It causes infections ranging from fatal pneumonia to asymptomatic carriage. Human infections with B. bronchiseptica are rare and typically limited to immunocompromised individuals. This species is considered the ancestral form from which the other two species, B. pertussis and B. parapertussis, have independently evolved through successive gene loss events. This reductive evolution is associated with their host adaptation and a more specialized pathogenic lifestyle in B. pertussis.
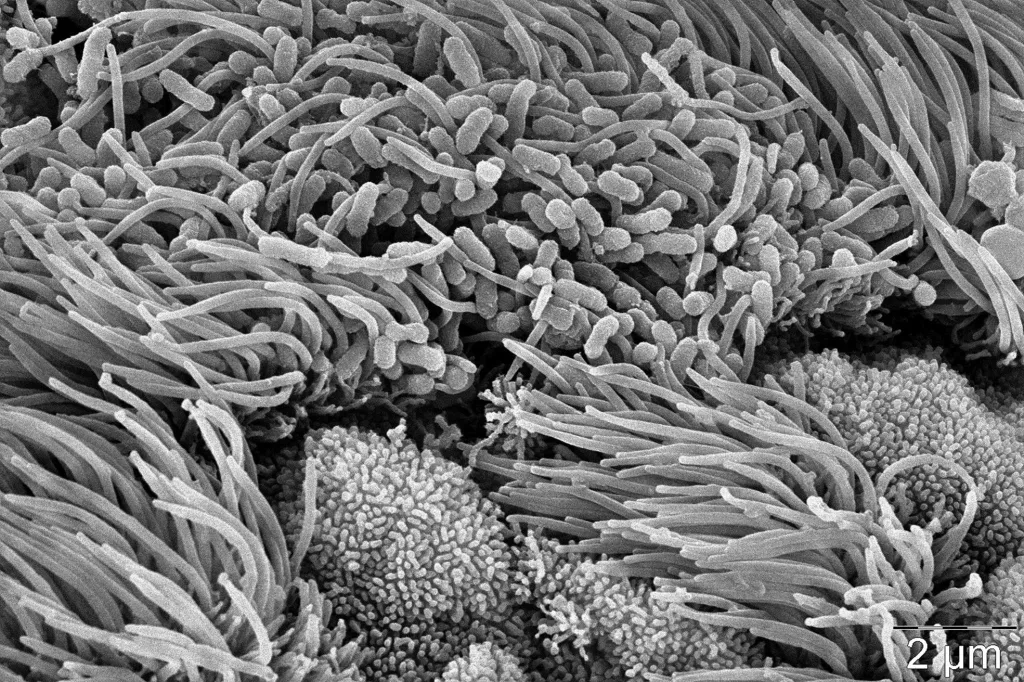
Colonization of the ciliated tracheal epithelial cells of mice trachea by B. pertussis. This figure was acquired in the collaboration with Dr. Olga Kofronova and Dr. Oldrich Benada at the Institute of Microbiology, Czech Academy of Sciences.
B. bronchiseptica and B. pertussis employ the T3SS injectisome to deliver the BteA effector into host cells. In this study, we visualized the needle tip filament of Bordetella T3SS injectisome, a structure formed by the Bsp22 protein. We demonstrate that during Bordetella cultivation in Stainer-Scholte medium, Bsp22 filaments are abundant and can dynamically extend up to several micrometers in length through the incorporation of new subunits at their distal ends. However, during infection of nasal epithelial cells (hNECs) in air-liquid interface cultures, Bsp22 filaments became scarce, with most bacteria lacking detectable filaments. These results highlight the adaptability of the Bordetella T3SS injectisome and show how its tip filament structure changes in response to different environments.

Malcova I, Zmuda M, Valecka J, and Kamanova J.* (2025). Assembly and dynamic regulation of the tip filament of the Bordetella type III secretion system injectisome. mBio, accepted. This work has been funded by grant 24-11053S of the Czech Science Foundation (www.gacr.cz), and a grant from the Programme Johannes Amos Comenius under the Ministry of Education, Youth and Sports of the Czech Republic (project Nr CZ.02.01.01/00/22_008/0004597).
The nasal epithelium is the initial site where B. pertussis comes into contact with the host during respiratory tract infection. In this study, human nasal epithelial cells (hNECs) cultured at the air-liquid interface were established as an in vitro model to investigate the early stages of B. pertussis infection. We showed that the clinical isolate B. pertussis B1917 resides in the mucus during the early stages of colonization without disrupting the epithelial barrier function. Infection results in moderate transcriptomic and proteomic changes, characterized by increased mucus production and minimal inflammatory signaling. These results suggest that B. pertussis B1917 may evade early host recognition by residing in mucus and avoiding direct interaction with epithelial cells. They also highlight the importance of other components of the mucosal immune system, such as resident immune cells, for the initiation of an effective defense.

Zmuda M, Malcova I, Pravdova B, Cerny O, Vondrova D, and Kamanova J.* (2025): Limited response of primary nasal epithelial cells to Bordetella pertussis infection. Microbiology Spectrum, https://doi.org/10.1128/spectrum.01267-25. This work was funded by the grant 21-05466S of the Czech Science Foundation (www.gacr.cz), a grant from the Programme Johannes Amos Comenius under the Ministry of Education, Youth and Sports of the Czech Republic (project Nr CZ.02.01.01/00/22_008/0004597), and the Lumina Queruntur Fellowship LQ200202001 of the Czech Academy of Sciences.
The respiratory pathogens B. pertussis and B. bronchiseptica exhibit cytotoxicity toward a variety of mammalian cells, which depends on the T3SS effector BteA. However, the molecular mechanism underlying BteA cytotoxicity is elusive. In this study, we performed a CRISPR-Cas9 screen, revealing that BteA-induced cell death depends on essential or redundant host processes. Additionally, we demonstrate that BteA disrupts calcium homeostasis, which leads to mitochondrial dysfunction and cell death.

Zmuda M, Sedlackova E, Pravdova B, Cizkova M, Dalecka M, Cerny O, Allsop TR, Grousl T, Malcova I, and Kamanova J.* (2024). The Bordetella effector protein BteA induces host cell death by disruption of calcium homeostasis, ZM and SE contributed equally, mBio, https://doi.org/10.1128/mbio.01925-24. This work was funded by the grant 21-05466S of the Czech Science Foundation (www.gacr.cz), a grant from the Programme Johannes Amos Comenius under the Ministry of Education, Youth and Sports of the Czech Republic (project Nr CZ.02.01.01/00/22_008/0004597), and the Lumina Queruntur Fellowship LQ200202001 of the Czech Academy of Sciences.
The T3SS is utilized by many Gram-negative bacteria to deliver effector proteins from bacterial cytosol directly into infected host cell cytoplasm in a regulated and targeted manner. Pathogenic bordetellae use the T3SS to inject the BteA and BopN proteins into infected cells and upregulate the production of the anti-inflammatory cytokine interleukin-10 (IL-10) to evade host immunity. Previous studies proposed that BopN acted as an effector in host cells. In this study, we report that BopN is a T3SS gatekeeper that regulates the secretion and translocation activity of Bordetella T3SS.

Navarrete MK, Bumba L, Prudnikova T, Malcova T, Romero Allsop T, Sebo P, and Kamanova J.* (2023): BopN is a gatekeeper of the Bordetella type III secretion system. Microbiology Spectrum, https://doi.org/10.1128/spectrum.04112-22. This work was funded by the grant 21-05466S of the Czech Science Foundation (www.gacr.cz) and the Lumina Queruntur Fellowship LQ200202001 of the Czech Academy of Sciences.
The T3SS effector protein BteA consists of two functional domains, an N-terminal lipid raft targeting (LRT) domain, and a cytotoxic C-terminal domain. In this study, we have described the lipid-binding specificity of the N-terminal LRT domain of BteA, which localizes to the plasma membrane through interaction with negatively charged phospholipids, including PIP2, PS and PA. A basic hydrophobic motif was identified that is essential for targeting to the membrane and shows homology to motifs found in unrelated bacterial toxins. The data also showed that the cytotoxicity of BteA is independent of LRT-mediated membrane localization.

Malcova I, Bumba L, Uljanic F, Kuzmenko D, Nedomova J., and Kamanova J* (2021), Lipid binding by the N-terminal motif mediates plasma membrane localization of Bordetella effector protein BteA. J Biol Chem 2021, 100607, IM and BL contributed equally, https://doi.org/10.1016/j.jbc.2021.100607. This work was funded by grant 18-16772Y of the Czech Science Foundation (www.gacr.cz) and Lumina Queruntur Fellowship LQ200202001 of the Czech Academy of Sciences.
Pertussis remains the least-controlled vaccine-preventable infectious disease and the mechanisms by which B. pertussis subverts defense mechanisms of human airway mucosa remain poorly understood. In this study, we found that B. pertussis had the cytotoxic activity of its T3SS-delivered effector BteA strongly attenuated by insertion of an alanine residue at position 503 as compared to the BteA homologue of the animal pathogen B. bronchiseptica. This functional adaptation reduced the capacity of B. pertussis to suppress host inflammatory response and may contribute to an acute course of the pulmonary form of human infant pertussis.

Bayram J, Malcova I, Sinkovec L, Holubova J, Streparola G, Jurnecka D, Kucera J, Sedlacek R, Sebo P and Kamanova J* (2020): Cytotoxicity of the effector protein BteA was attenuated in Bordetella pertussis by insertion of an alanine residue. PLoS Pathog. 16(8):e1008512, https://doi.org/10.1371/journal.ppat.1008512. This work was funded by grant 18-16772Y of the Czech Science Foundation (www.gacr.cz).